CO2-free hydrogen production from methane
Sciencedaily - H2 from methane without CO2 emission
Tool to calculate CO2 emissions from fossil fuel combustion
hydrogen production via the direct cracking of methane over Ni/SiO2
Hydrogen production from natural gas
Liquid hydrogen production via hydrogen sulfide methane reformation
The production of hydrogen from methane using tubular plasma reactors
Catalysts for splitting water
When using sunlight to split water, we need a catalyst. However, the cost of an ounce of platinum (the most efficient catalyst for that matter) is about $2,000. Other metals work but aren't nearly as efficient as platinum. This article talks about the use of catalysts for splitting water with sunlight, and how to make it efficient.
http://www.rsc.org/chemistryworld/News/2008/July/31070802.asp
http://www.rsc.org/chemistryworld/News/2008/July/31070802.asp
Changes in global temperature
Even though that's not the main focus of my research, my topic definitely overlaps with common environmental issues, such as air pollution. The increase in temperature is a consequence of many actions, which include air pollution. As I stated previously, about 45% of the air pollution is caused by vehicles.
http://www.metoffice.gov.uk/hadobs/hadcrut3/HadCRUT3_accepted.pdf
http://www.metoffice.gov.uk/hadobs/hadcrut3/HadCRUT3_accepted.pdf
MIT group uses cobalt-based catalyst and sunlight to split water
Reactive metals can be used as catalysts to decrease the activation energy of a reaction. Since the H hydrogen bonds to O, it forms a strong bond. Simply using sunlight would not be enough to break this bond, because there isn't enough energy in sunlight to break the intramolecular bonds, the water would simply get heated up. However, a group of MIT researches developed an artificial leaf that uses a cobalt-based catalyst to make sunlight a viable option for breaking the O-H bond.
| nocera_artificail_leaf.pdf | |
| File Size: | 616 kb |
| File Type: | |
University of Colorado uses redox cycle for splitting water
| H2 Generation | |
| File Size: | 1236 kb |
| File Type: | |
Photodissociation of water vapor
| brinkman_1969.pdf | |
| File Size: | 4153 kb |
| File Type: | |
High-pressure photodissociation of water
| high_pressure_photodissociation.pdf | |
| File Size: | 1479 kb |
| File Type: | |
Hydrogen Fuel Cell
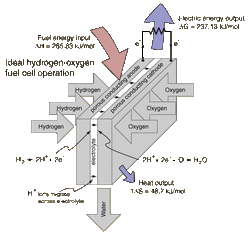
Hydrogen and oxygen can be combined in a fuel cell to produce electrical energy. A fuel cell uses a chemical reaction to provide an external voltage, as does a battery, but differs from a battery in that the fuel is continually supplied in the form of hydrogen and oxygen gas. It can produce electrical energy at a higher efficiency than just burning the hydrogen to produce heat to drive a generator because it is not subject to the thermal bottleneck from the second law of thermodynamics. It's only product is water, so it is pollution-free. All these features have led to periodic great excitement about its potential, but we are still in the process of developing that potential as a pollution-free, efficient energy source (see Kartha and Grimes).
Combining a mole of hydrogen gas and a half-mole of oxygen gas from their normal diatomic forms produces a mole of water. A detailed analysis of the process makes use of the thermodynamic potentials. This process is presumed to be at 298K and one atmosphere pressure, and the relevant values are taken from a table of thermodynamic properties.
Quantity H2 0.5 O2 H2O Change
Enthalpy 0 0 -285.83 kJ ΔH = -285.83 kJ
Entropy 130.68 J/K 0.5 x 205.14 J/K 69.91 J/K TΔS = -48.7 kJ
Energy is provided by the combining of the atoms and from the decrease of the volume of the gases. Both of those are included in the change in enthalpy included in the table above. At temperature 298K and one atmosphere pressure, the system work is
W = PΔV = (101.3 x 103 Pa)(1.5 moles)(-22.4 x 10-3 m3/mol)(298K/273K) = -3715 J
Since the enthalpy H= U+PV, the change in internal energy U is then
ΔU = ΔH - PΔV = -285.83 kJ - 3.72 kJ = -282.1 kJ
The entropy of the gases decreases by 48.7 kJ in the process of combination since the number of water molecules is less than the number of hydrogen and oxygen molecules combining. Since the total entropy will not decrease in the reaction, the excess entropy in the amount TΔS must be expelled to the environment as heat at temperature T. The amount of energy per mole of hydrogen which can be provided as electrical energy is the change in the Gibbs free energy:
ΔG = ΔH - TΔS = -285.83 kJ + 48.7 kJ = -237.1 kJ
For this ideal case, the fuel energy is converted to electrical energy at an efficiency of 237.1/285.8 x100% = 83%! This is far greater than the ideal efficiency of a generating facility which burned the hydrogen and used the heat to power a generator! Although real fuel cells do not approach that ideal efficiency, they are still much more efficient than any electric power plant which burns a fuel.
Comparison of electrolysis and the fuel cell process
In comparing the fuel cell process to its reverse reaction, electrolysis of water, it is useful treat the enthalpy change as the overall energy change. The Gibbs free energy is that which you actually have to supply if you want to drive a reaction, or the amount that you can actually get out if the reaction is working for you. So in the electrolysis/fuel cell pair where the enthalpy change is 285.8 kJ, you have to put in 237 kJ of energy to drive electrolysis and the heat from the environment will contribute TΔS=48.7 kJ to help you. Going the other way in the fuel cell, you can get out the 237 kJ as electric energy, but have to dump TΔS = 48.7 kJ to the environment.
From http://hyperphysics.phy-astr.gsu.edu/
Combining a mole of hydrogen gas and a half-mole of oxygen gas from their normal diatomic forms produces a mole of water. A detailed analysis of the process makes use of the thermodynamic potentials. This process is presumed to be at 298K and one atmosphere pressure, and the relevant values are taken from a table of thermodynamic properties.
Quantity H2 0.5 O2 H2O Change
Enthalpy 0 0 -285.83 kJ ΔH = -285.83 kJ
Entropy 130.68 J/K 0.5 x 205.14 J/K 69.91 J/K TΔS = -48.7 kJ
Energy is provided by the combining of the atoms and from the decrease of the volume of the gases. Both of those are included in the change in enthalpy included in the table above. At temperature 298K and one atmosphere pressure, the system work is
W = PΔV = (101.3 x 103 Pa)(1.5 moles)(-22.4 x 10-3 m3/mol)(298K/273K) = -3715 J
Since the enthalpy H= U+PV, the change in internal energy U is then
ΔU = ΔH - PΔV = -285.83 kJ - 3.72 kJ = -282.1 kJ
The entropy of the gases decreases by 48.7 kJ in the process of combination since the number of water molecules is less than the number of hydrogen and oxygen molecules combining. Since the total entropy will not decrease in the reaction, the excess entropy in the amount TΔS must be expelled to the environment as heat at temperature T. The amount of energy per mole of hydrogen which can be provided as electrical energy is the change in the Gibbs free energy:
ΔG = ΔH - TΔS = -285.83 kJ + 48.7 kJ = -237.1 kJ
For this ideal case, the fuel energy is converted to electrical energy at an efficiency of 237.1/285.8 x100% = 83%! This is far greater than the ideal efficiency of a generating facility which burned the hydrogen and used the heat to power a generator! Although real fuel cells do not approach that ideal efficiency, they are still much more efficient than any electric power plant which burns a fuel.
Comparison of electrolysis and the fuel cell process
In comparing the fuel cell process to its reverse reaction, electrolysis of water, it is useful treat the enthalpy change as the overall energy change. The Gibbs free energy is that which you actually have to supply if you want to drive a reaction, or the amount that you can actually get out if the reaction is working for you. So in the electrolysis/fuel cell pair where the enthalpy change is 285.8 kJ, you have to put in 237 kJ of energy to drive electrolysis and the heat from the environment will contribute TΔS=48.7 kJ to help you. Going the other way in the fuel cell, you can get out the 237 kJ as electric energy, but have to dump TΔS = 48.7 kJ to the environment.
From http://hyperphysics.phy-astr.gsu.edu/
Electrolysis of Water
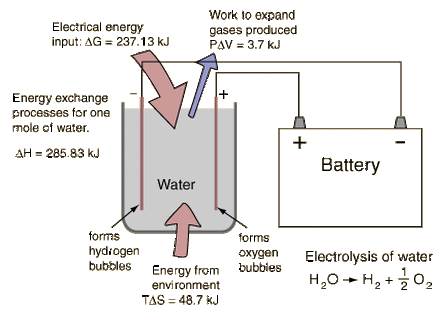
By providing energy from a battery, water (H2O) can be dissociated into the diatomic molecules of hydrogen (H2) and oxygen (O2). This process is a good example of the the application of the four thermodynamic potentials.
The electrolysis of one mole of water produces a mole of hydrogen gas and a half-mole of oxygen gas in their normal diatomic forms. A detailed analysis of the process makes use of the thermodyamic potentials and the first law of thermodynamics. This process is presumed to be at 298K and one atmosphere pressure, and the relevant values are taken from a table of thermodynamic properties.
Quantity H2O H2 0.5 O2 Change
Enthalpy -285.83kJ 0 0 ΔH = 285.83 kJ
Entropy 69.91 J/K 130.68 J/K 0.5 x 205.14 J/K TΔS = 48.7 kJ
The process must provide the energy for the dissociation plus the energy to expand the produced gases. Both of those are included in the change in enthalpy included in the table above. At temperature 298K and one atmosphere pressure, the system work is
W = PΔV = (101.3 x 103 Pa)(1.5 moles)(22.4 x 10-3 m3/mol)(298K/273K) = 3715 J
Since the enthalpy H= U+PV, the change in internal energy U is then
ΔU = ΔH - PΔV = 285.83 kJ - 3.72 kJ = 282.1 kJ
This change in internal energy must be accompanied by the expansion of the gases produced, so the change in enthalpy represents the necessary energy to accomplish the electrolysis. However, it is not necessary to put in this whole amount in the form of electrical energy. Since the entropy increases in the process of dissociation, the amount TΔS can be provided from the environment at temperature T. The amount which must be supplied by the battery is actually the change in the Gibbs free energy:
ΔG = ΔH - TΔS = 285.83 kJ - 48.7 kJ = 237.1 kJ
Since the electrolysis process results in an increase in entropy, the environment "helps" the process by contributing the amount TΔS. The utility of the Gibbs free energy is that it tells you what amount of energy in other forms must be supplied to get the process to proceed.
From http://hyperphysics.phy-astr.gsu.edu/
The electrolysis of one mole of water produces a mole of hydrogen gas and a half-mole of oxygen gas in their normal diatomic forms. A detailed analysis of the process makes use of the thermodyamic potentials and the first law of thermodynamics. This process is presumed to be at 298K and one atmosphere pressure, and the relevant values are taken from a table of thermodynamic properties.
Quantity H2O H2 0.5 O2 Change
Enthalpy -285.83kJ 0 0 ΔH = 285.83 kJ
Entropy 69.91 J/K 130.68 J/K 0.5 x 205.14 J/K TΔS = 48.7 kJ
The process must provide the energy for the dissociation plus the energy to expand the produced gases. Both of those are included in the change in enthalpy included in the table above. At temperature 298K and one atmosphere pressure, the system work is
W = PΔV = (101.3 x 103 Pa)(1.5 moles)(22.4 x 10-3 m3/mol)(298K/273K) = 3715 J
Since the enthalpy H= U+PV, the change in internal energy U is then
ΔU = ΔH - PΔV = 285.83 kJ - 3.72 kJ = 282.1 kJ
This change in internal energy must be accompanied by the expansion of the gases produced, so the change in enthalpy represents the necessary energy to accomplish the electrolysis. However, it is not necessary to put in this whole amount in the form of electrical energy. Since the entropy increases in the process of dissociation, the amount TΔS can be provided from the environment at temperature T. The amount which must be supplied by the battery is actually the change in the Gibbs free energy:
ΔG = ΔH - TΔS = 285.83 kJ - 48.7 kJ = 237.1 kJ
Since the electrolysis process results in an increase in entropy, the environment "helps" the process by contributing the amount TΔS. The utility of the Gibbs free energy is that it tells you what amount of energy in other forms must be supplied to get the process to proceed.
From http://hyperphysics.phy-astr.gsu.edu/
